Importance of Raw Material Testing for Pharmaceuticals
Raw material testing is crucially important for ensuring safety, quality and efficacy of pharmaceutical products.
There are many things to be considered that could impact the way raw materials need to be blended, such as polymorphism, the particle size of raw materials and other properties.
Hence, raw material analysis is essential to determine the purity, identity and quality of the raw materials before they go into the manufacturing process.
Need for Raw Material Testing
As hundreds of raw materials and ingredients are used in the process of formulating the final pharmaceutical product, it is quite tough to check every ingredient for quality.
Unless the ingredients have undergone quality testing, beginning the manufacturing process won’t be possible. Moreover, if low-quality raw materials are used, it will result in a low-quality finished product which could face product recall.
This can cause significant damage to material costs as well as reputation. Therefore, raw material testing in pharmaceuticals is necessary.
Ensuring a Quality product
Pharmaceutical raw material testing is carried out to establish that all the incoming raw materials match the right specifications and requirements. Needless to say, incorrect supply of raw materials will lead to a compromise in the safety and quality of the end product.
Besides, it will also cause manufacturing delays and significant wastage of time and costs. Therefore, testing labs help pharmaceutical companies lay down the specifications for the raw materials right from the initial stages of drug development.
Testing Labs: Standards and approvals needed
Every pharmaceutical product or medical device has to be approved by the State FDA and the Central Drugs Standards Control Organisation (CDSCO) before it is rolled out for public use or commercialization.
Testing laboratories can help carry out material analysis, DSC analysis, chemical tests, physical characterisation, NMR testing, FTIR testing and more, all according to the specifications and safety protocols established by the FDA and CDSCO.
Traditionally, chemical testing laboratories perform the raw material testing and prepare the reports to determine their quality and suitability to be used in pharmaceutical drug formulations. They are well equipped to carry out the sophisticated procedures involved in raw material tests.
Raw Materials Testing at Arbro
We are the leading pharmaceutical testing lab in India with over 25 years of experience in the analysis of raw materials and finished products. We offer a complete solution for testing of pharmaceutical raw materials including pharmaceutical drug substances, intermediates, excipients et cetera.
Our years of experience in chemical testing, physical characterization, and microbiological analysis as per pharmacopoeial requirements help you face these challenges of capacities and capabilities.
Given below is a list of some of the most common test that we carry out for pharmaceutical raw materials using either pharmacopoeial, client or in-house methods –
- Assay
- Impurities and Related Substances
- Residual Solvents and Organic Volatile Impurities
- Identification tests by FTIR, chemical analysis et cetera
- Limit tests for heavy metals by chemical methods or by ICP or ICP MS as per US 232, 233
- Microbial limit tests
- Sterility testing
- Microbiological assays
- Particle size distribution by optical microscopy
- Particle size distribution by laser diffraction particle size analyser – Malvern
- Test for crystallinity
- Melting point
- Differential scanning calorimetry (DSC)
- X-ray diffraction (XRD)
- Nuclear Magnetic Resonance (NMR)
- and many more tests
The pharmaceutical industry uses a very large variety of raw materials and literally hundreds of ingredients can go into the manufacturing of a single product. It is often difficult if not impossible for a pharmaceutical manufacturer to have in-house testing facilities and capabilities for all the ingredients and products.
Even if all the facilities are available in-house invariably there is a pressure on the capacity available and it is essential to have a trusted partner who can carry out the testing of pharmaceutical raw materials before they can be released and used for manufacturing of the pharmaceutical products.
Besides routine analysis for release testing of pharmaceutical raw materials we also carry out method validation and development studies for all of these tests.
Our laboratories have also supported numerous manufacturers of pharmaceutical raw materials for carrying out stability is for their pharmaceutical drug substances and excipients.
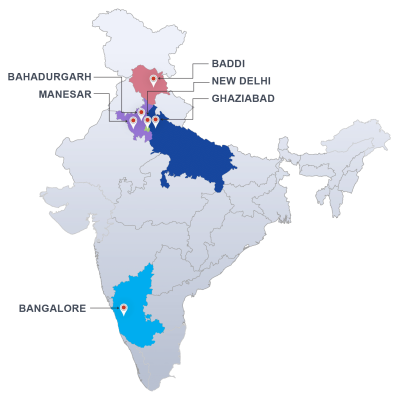
Our laboratories are approved by the State Food and Drug Administration (FDA) and also by the Central Drugs Standards Control Organisation and we can provide reports on Form 39 as per the requirements of the Drugs and Cosmetics Act. We have approved laboratories in New Delhi, Himachal Pradesh and Bangalore.
Our laboratories in Delhi and Himachal Pradesh are also accredited by the National accreditation board for testing and calibration laboratories to the ISO standard 17025.
You can contact us now using the quick query form on the right or you can call us directly on +91 – 11 – 45754575 and our team will be happy to provide you a quotation and proposal for your pharmaceutical raw material testing requirements.
FAQs
1. What is raw material testing?
Before manufacturing a drug or food product, each of its raw material is tested individually. This is called raw material testing, and it is compulsory as per the terms of the FDA to establish the materials’ identity, purity, safety, and quality. These aspects of pharmaceutical elements make sure that the end product delivers what it is intended to.
2. What is the sample quantity needed for typical raw material analysis?
Usually, raw material tests require enough portion of the sample to carry out all the anticipated test procedures. Repetition and retention samples must be included in this. So the exact quantity is determined based on the tests to be conducted.
3. Do you have HPLC for assay and impurity tests?
Yes, our state-of-the-art laboratory consists of proper equipment for chromatography analytical methodologies, such as TLC, HPLC, GC, HPTLC, etc. for pharmaceutical raw material testing.
4. Do you have FTIR for raw material identification?
Along with chromatography equipment, our laboratory also has all spectroscopy tools for conducting FTIR, Atomic Absorption, and UV/visible.