Grow with Us!
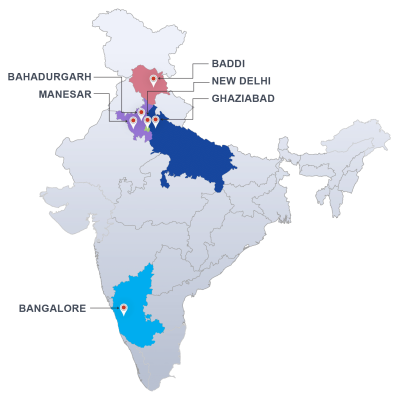
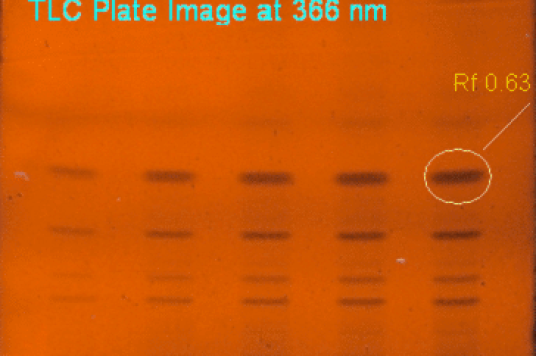
We were the first Herbal Testing Lab in India to be Licensed by Directorate of ISM & H and are recognized by AYUSH and accredited by NABL ISO/IEC 17025. We carry out testing of herbal products including the testing of raw materials, excipients and finished single herb as well as poly herbal formulations.