Grow With Us!
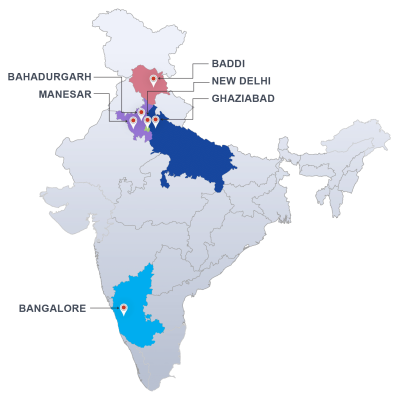
Our state of the art laboratories in pan India locations offers pharmaceutical testing and research services to support the development , approval and manufacture of pharmaceutical raw materials and finished products. Our cGMP and GLP compliance ensures that we fulfill international regulatory standards and commitment to provide excellent quality products and services.